2.2 The Development of the Atomic Model
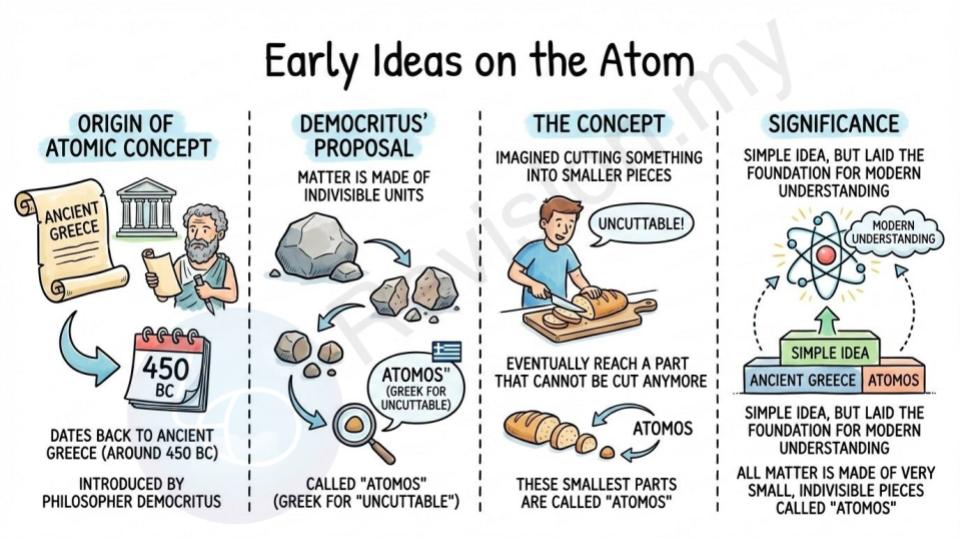
Early Ideas on the Atom
Origin of atomic concept: The idea that everything is made up of tiny building blocks called atoms started a long time ago in ancient Greece. Around 450 BC, a philosopher named Democritus came up with this idea. He imagined that if you kept cutting something into smaller and smaller pieces, you would eventually get to a part that could not be cut anymore. This smallest part, he called an “atomos,” which means “uncuttable” in Greek.
Democritus’ proposal: Democritus believed that all matter (everything around us) is made up of very small, indivisible pieces called ‘atomos.’ He said these pieces were so tiny that they couldn’t be divided any further. This idea was very simple, but it laid the foundation for our modern understanding of atoms.
Key Models of the Atom
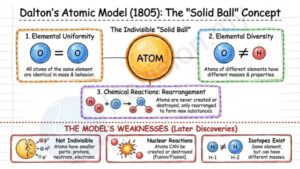
John Dalton's Atomic Model (1805)
Indivisible particles: Dalton was a scientist who said that atoms are the smallest parts of matter and cannot be broken into anything smaller. He believed atoms were like solid balls that couldn’t be split apart.
Elemental uniformity: Dalton also said that all atoms of the same element are exactly the same—they have the same mass and behave in the same way.
Elemental diversity: But atoms of different elements are not the same. For example, an atom of oxygen is not the same as an atom of hydrogen—they have different weights and properties.
Conservation in reactions: Dalton explained that in chemical reactions, atoms are never created, destroyed, or split. They simply rearrange to make new substances.
Combination of atoms: According to Dalton, chemical reactions happen when atoms stick together or change partners to form new materials.
Model depiction: He imagined atoms as small, solid balls—like tiny marbles—that can’t be broken down.
Weakness of Dalton’s Model:
- Atoms consist of even smaller particles called electrons, protons and neutrons.
- Atoms can be created and destroyed in the nuclear reactions such as nuclear fusion and nuclear fission.
- Atoms of the same element can have different physical properties, for example, isotopes of hydrogen.
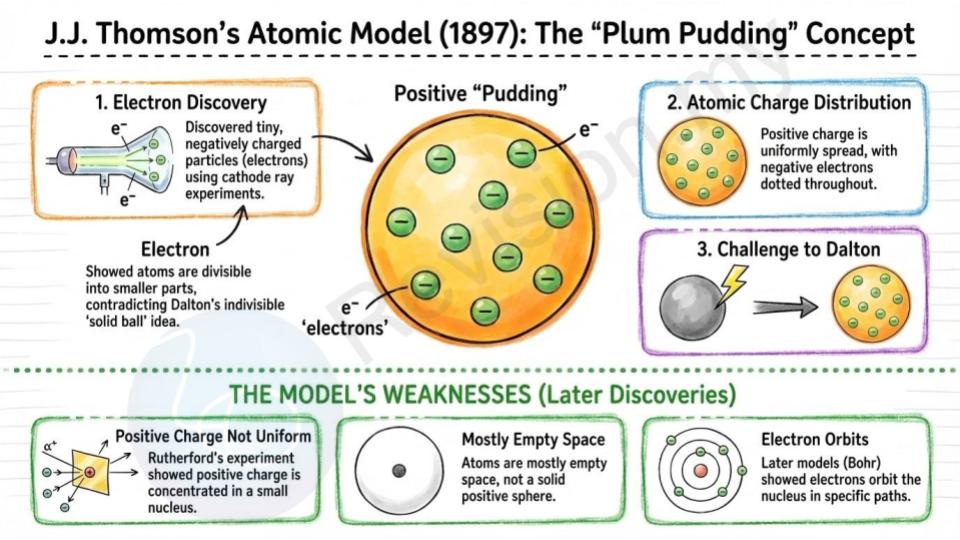
J.J. Thomson's Atomic Model (1897)
Electron discovery: Thomson discovered that atoms are not solid after all. He used a special experiment with cathode rays and found out that atoms have tiny, negatively charged particles called electrons.
Plum pudding model: To explain this, Thomson came up with a new model. He said an atom is like a pudding with raisins in it. The pudding is a positively charged ball, and the electrons (raisins) are scattered inside it.
Atomic charge distribution: In his model, the positive charge is spread out all over the atom, and the small negative electrons are dotted throughout the positive background.
Challenge to Dalton: This was a big change from Dalton’s idea, because it showed that atoms are made of even smaller parts and are not indivisible after all.
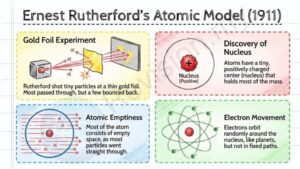
Ernest Rutherford's Atomic Model (1911)
Gold foil experiment: Rutherford did a famous experiment where he shot tiny particles at a very thin sheet of gold foil. Most particles passed through, but a few bounced back.
Discovery of nucleus: From this, he figured out that atoms have a small center, called the nucleus, which has a positive charge and holds most of the atom’s mass.
Atomic emptiness: Rutherford found that atoms are mostly empty space, because most of the particles went straight through the foil.
Electron movement: He said electrons move around the nucleus, kind of like planets orbiting the sun, but not in fixed paths yet.
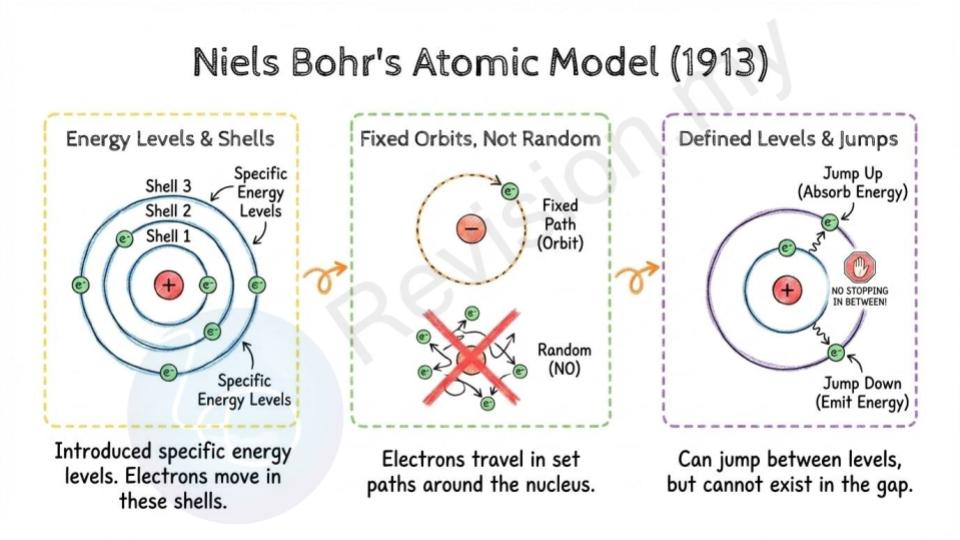
Niels Bohr's Atomic Model (1913)
Electron energy levels: Bohr improved Rutherford’s model by suggesting that electrons move in specific energy levels, also called shells.
Fixed electron orbits: He said electrons don’t just zoom around randomly. They travel in fixed paths or orbits around the nucleus.
Energy level restriction: Each electron can only stay in certain allowed orbits. They can jump between these orbits, but they can’t be in between them.
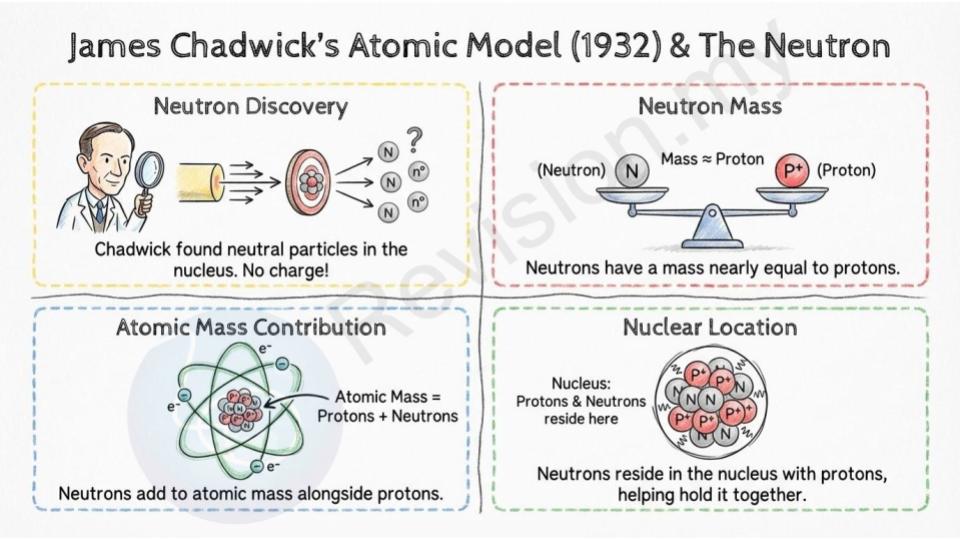
James Chadwick's Atomic Model (1932)
Neutron discovery: Chadwick discovered another important particle inside the atom—the neutron. Neutrons don’t have any charge.
Neutron mass: He found out that neutrons are about the same weight as protons, the positively charged particles in the nucleus.
Atomic mass contribution: Neutrons, along with protons, add to the weight of the atom.
Nuclear location: Neutrons live inside the nucleus with the protons, helping to hold it together.